
Green synthesis, characterization and catalytic degradation studies of gold nanoparticles against congo red and methyl orange - ScienceDirect

Degradation of methylene blue and methyl orange by palladium-doped TiO2 photocatalysis for water reuse: Efficiency and degradation pathways - ScienceDirect
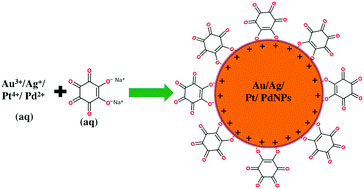
Green synthesis of gold, silver, platinum, and palladium nanoparticles reduced and stabilized by sodium rhodizonate and their catalytic reduction of 4-nitrophenol and methyl orange - New Journal of Chemistry (RSC Publishing)

Complex formation reactions of palladium(II)-1,3-diaminopropane with various biologically relevant ligands. Kinetics of hydrolysis of glycine methyl ester through complex formation – topic of research paper in Chemical sciences. Download scholarly ...

Polyaniline Supported Palladium Catalyzed Reductive Degradation of Dyes Under Mild Condition | Bentham Science

Highly photostable palladium-loaded TiO2 nanotubes and the active species in the photodegradation of methyl orange - ScienceDirect
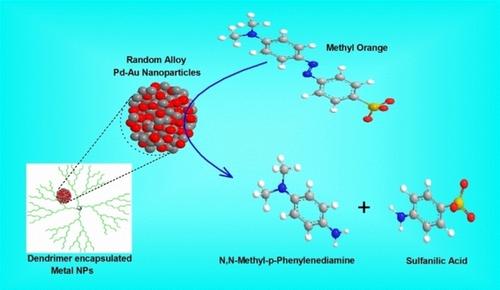
Effective Catalytic Reduction of Methyl Orange Catalyzed by the Encapsulated Random Alloy Palladium‐Gold Nanoparticles Dendrimer. - ChemistrySelect - X-MOL

Figure 2 from Rapid degradation of azo dye methyl orange using hollow cobalt nanoparticles. | Semantic Scholar

Room-temperature synthesis of air stable cobalt nanoparticles and their use as catalyst for methyl orange dye degradation - ScienceDirect
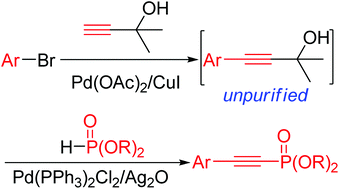
Palladium-catalyzed oxidative deacetonative coupling of 4-aryl-2-methyl-3-butyn-2-ols with H-phosphonates - Organic & Biomolecular Chemistry (RSC Publishing)
Catalyzed oxidative degradation of methyl orange over Au catalyst prepared by ionic liquid-polymer modified silica

Materials | Free Full-Text | Palladium/Carbon Nanofibers by Combining Atomic Layer Deposition and Electrospinning for Organic Pollutant Degradation | HTML

China CAS 12081-22-0 (1-Methylallyl) Palladium Chloride Dimer C8h14cl2pd2 - China Palladium Catalyst, Ruthenium Catalyst
Effective Catalytic Reduction of Methyl Orange Catalyzed by the Encapsulated Random Alloy Palladiumв•'Gold Nanoparticles De

Catalysts | Free Full-Text | Polyaniline-Grafted RuO2-TiO2 Heterostructure for the Catalysed Degradation of Methyl Orange in Darkness
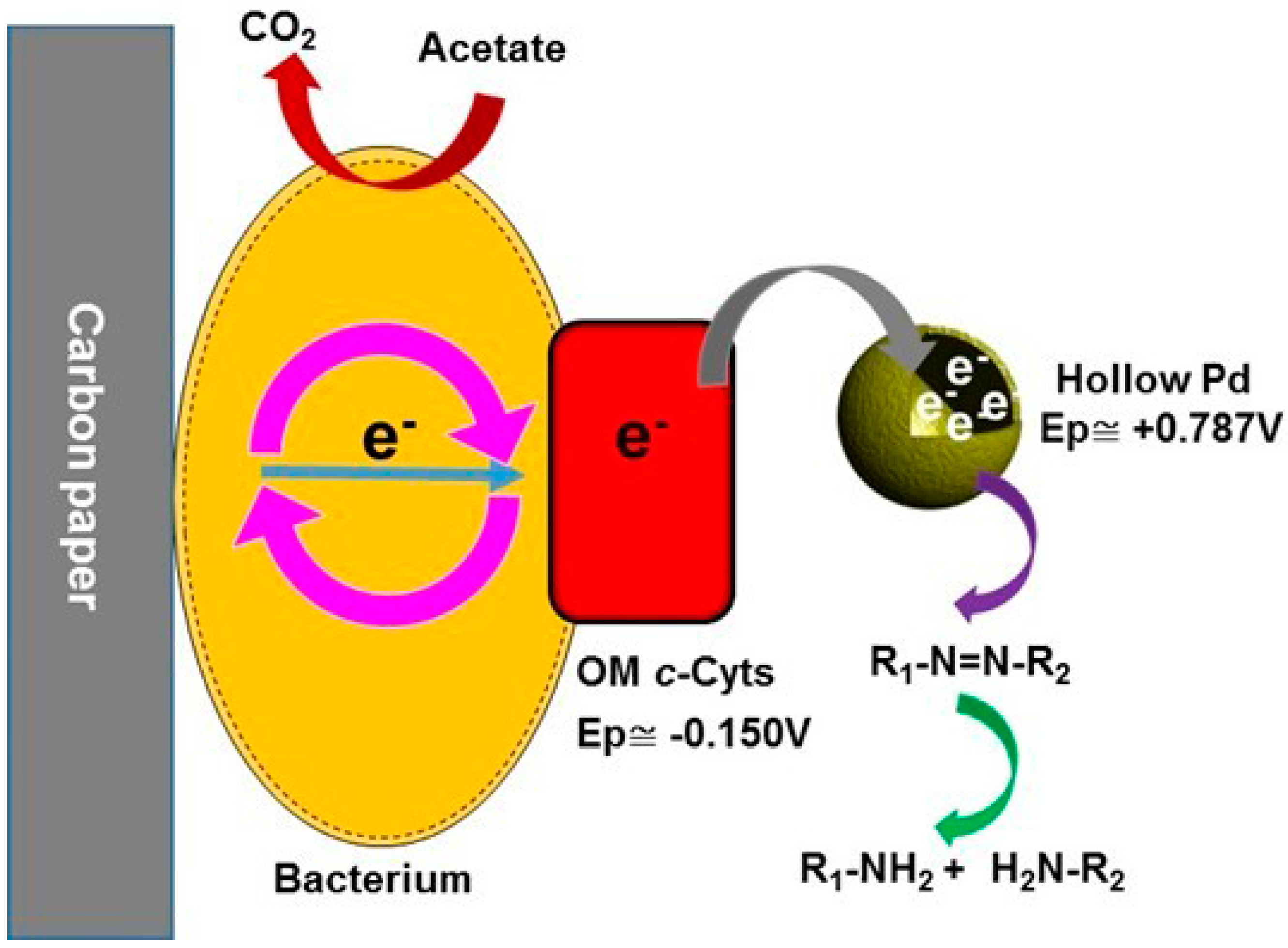
Materials | Free Full-Text | Hollow Palladium Nanoparticles Facilitated Biodegradation of an Azo Dye by Electrically Active Biofilms | HTML

Figure 3 from Rapid degradation of azo dye methyl orange using hollow cobalt nanoparticles. | Semantic Scholar

Inorganic self-assembly through sequential complexation in the formation of bimetallic and trimetallic architectures from multisite ligands based on 5,5'-disubstituted 2,2'-bipyridines



