
Graphene anchored palladium complex as efficient and recyclable catalyst in the Heck cross-coupling reaction - ScienceDirect
Spectrophotometric determination of the platinum metals VI.* Determination of rhodium and palladium with 4-(2-thiazolylazo)resor
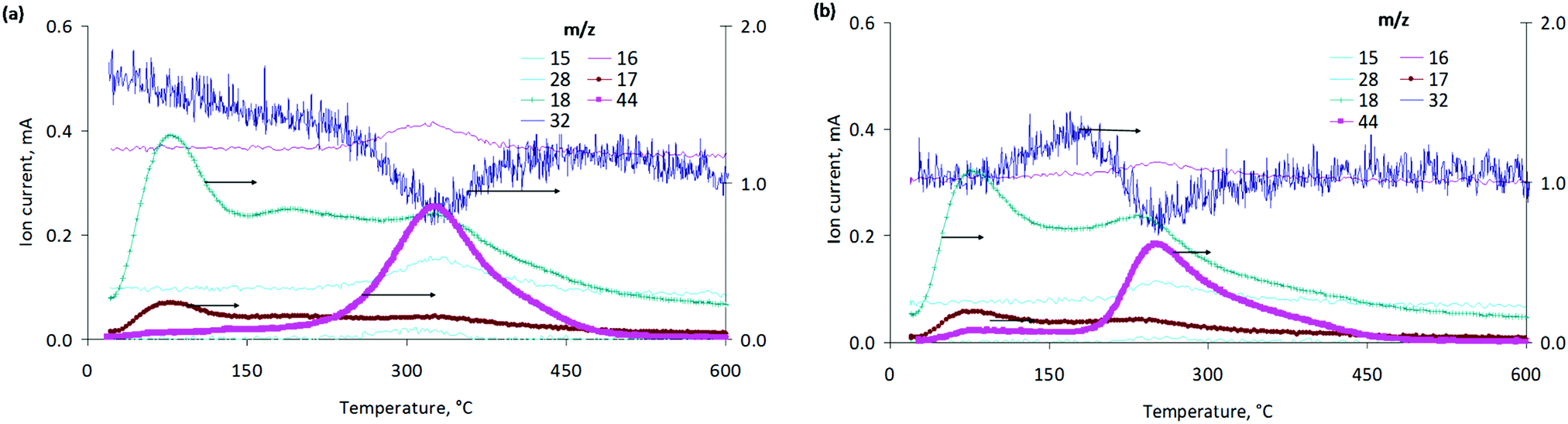
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)

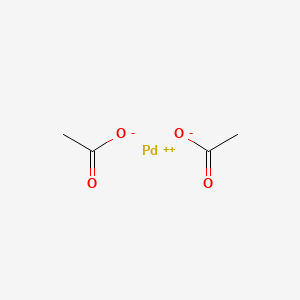
Spectra of (a) 0.05 mM Pd(COO(CH 3 )) 2 and (b) 0.01 mg cm − 3 Pd par- | Download Scientific Diagram

Graphene anchored palladium complex as efficient and recyclable catalyst in the Heck cross-coupling reaction - ScienceDirect
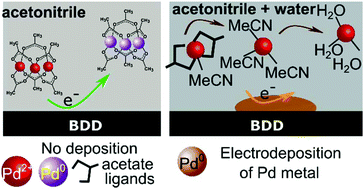
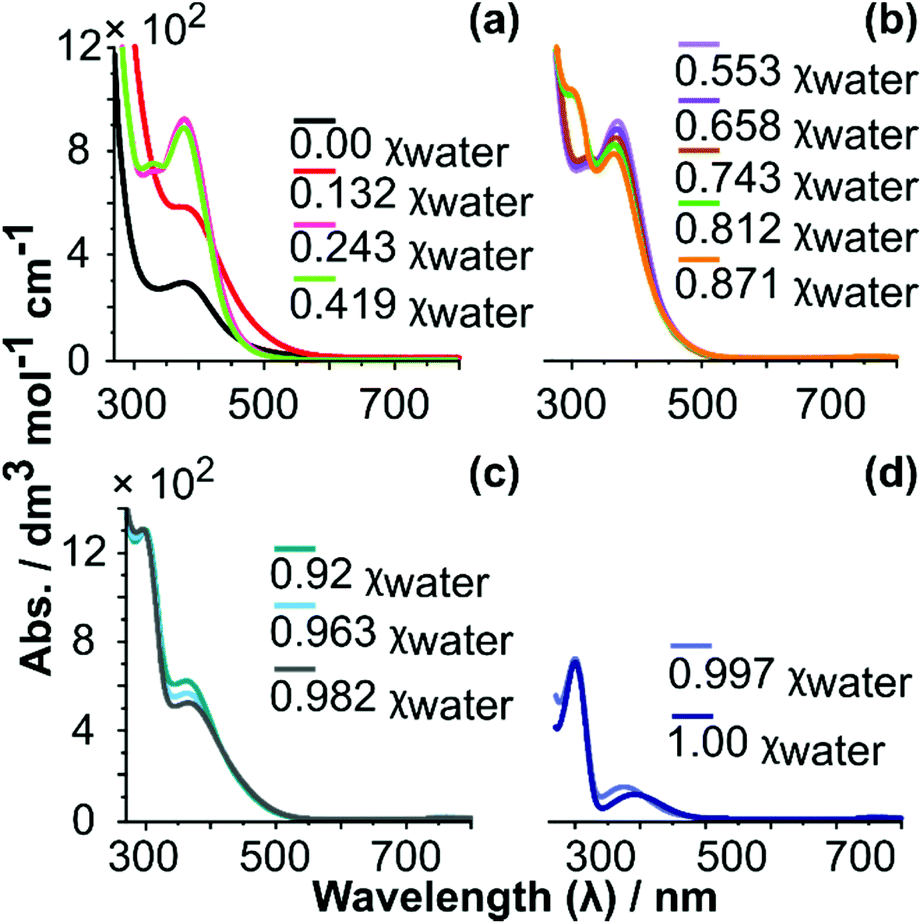
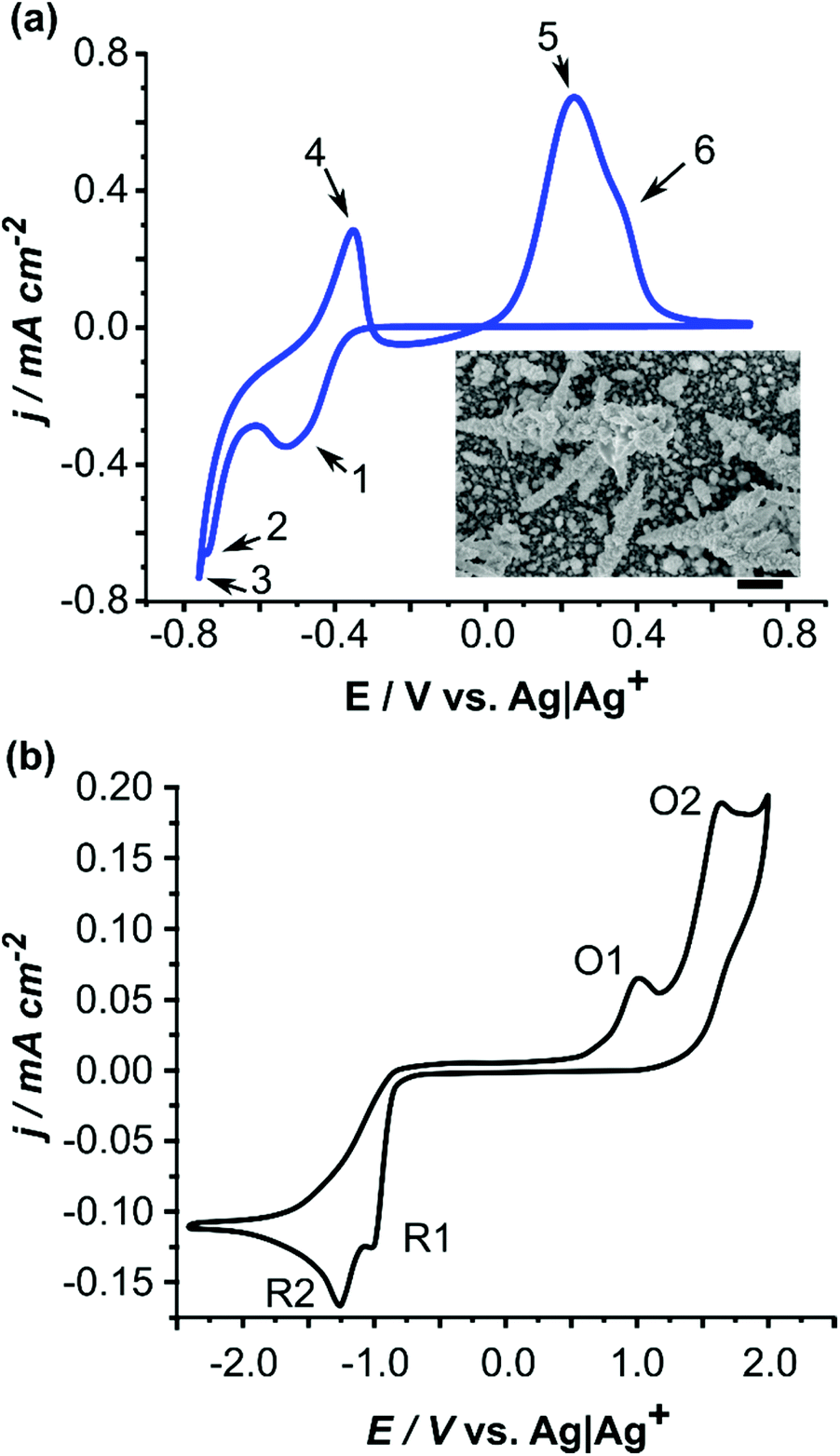
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)

Ligand characteristics and in situ generation of Pd active species towards CC coupling using series of 2-(1H-imidazol-2-yl)phenols - ScienceDirect
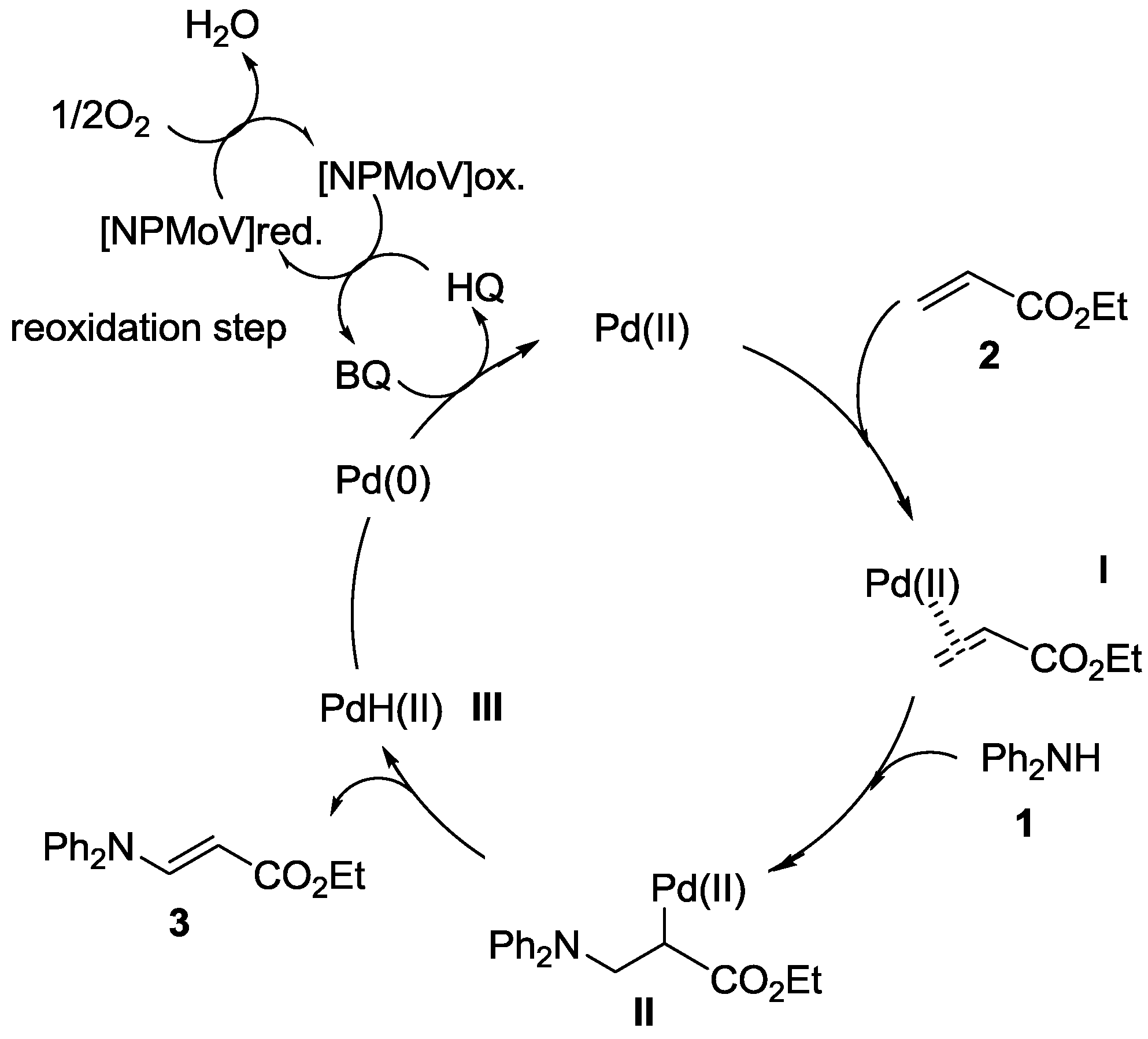
Catalysts | Free Full-Text | Palladium-Catalyzed Intermolecular Oxidative Amination of Alkenes with Amines, Using Molecular Oxygen as Terminal Oxidant | HTML

Activating electrochemical catalytic activity of bio-palladium by hybridizing with carbon nanotube as “e − Bridge” | Scientific Reports

EIS of the bare GCE and Pd/CNTs-Nafion modified electrodes in 0.1 M KCl... | Download Scientific Diagram
Synthesis and characterisation of palladium(II) complexes with new diphosphine ligands. Application in the alternating copolymer

PDF) Controlling palladium morphology in electrodeposition from nanoparticles to dendrites via the use of mixed solvents

Palladium and palladium–tin supported on multi wall carbon nanotubes or carbon for alkaline direct ethanol fuel cell - ScienceDirect
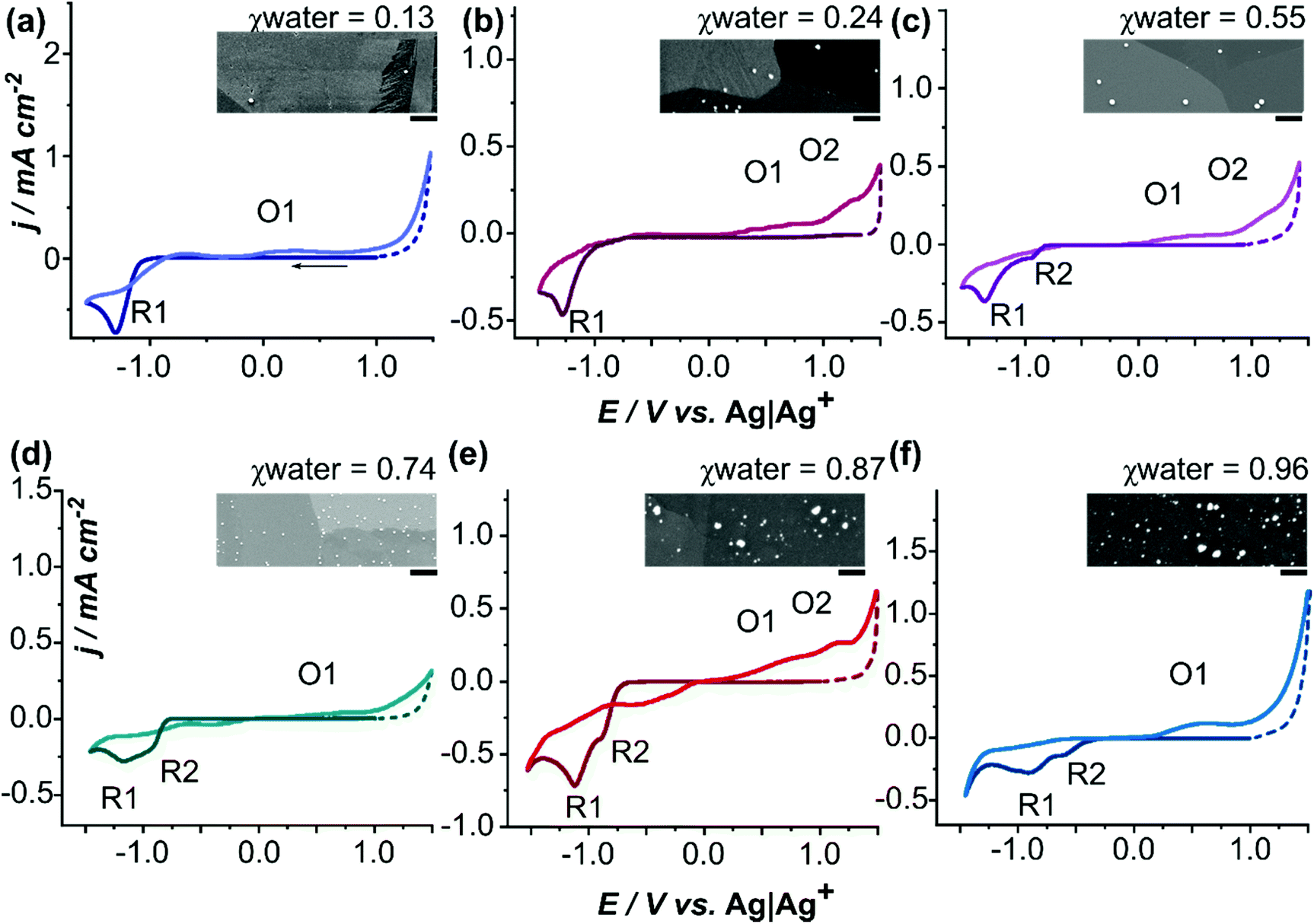
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)
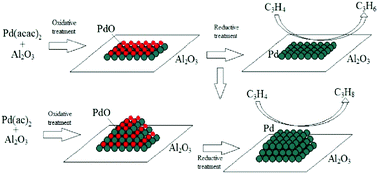
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)

Global transcriptional analysis of Geobacter sulfurreducens under palladium reducing conditions reveals new key cytochromes involved | SpringerLink
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)
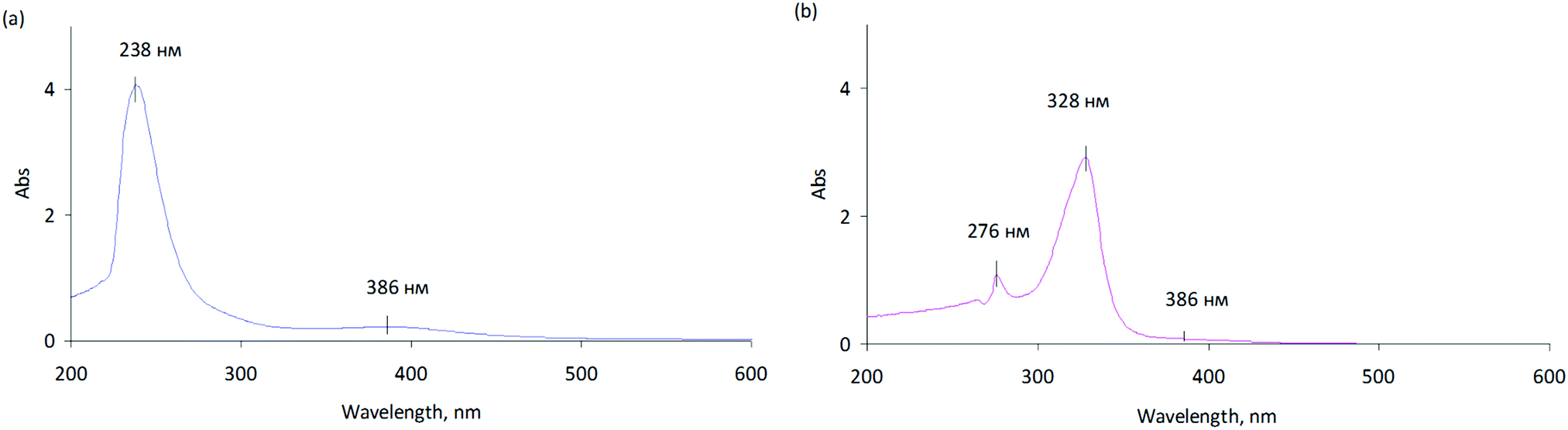
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)
![Plot between rate of reaction (-dc/dt) vs [H + ] and [KCl] at T = 303K.... | Download Scientific Diagram Plot between rate of reaction (-dc/dt) vs [H + ] and [KCl] at T = 303K.... | Download Scientific Diagram](https://www.researchgate.net/profile/Ajaya_Singh2/publication/240976537/figure/fig3/AS:669005698318346@1536514796550/Fig-3-Plot-between-rate-of-reaction-dc-dt-vs-H-and-KCl-at-T-303K-NBP.png)
Plot between rate of reaction (-dc/dt) vs [H + ] and [KCl] at T = 303K.... | Download Scientific Diagram
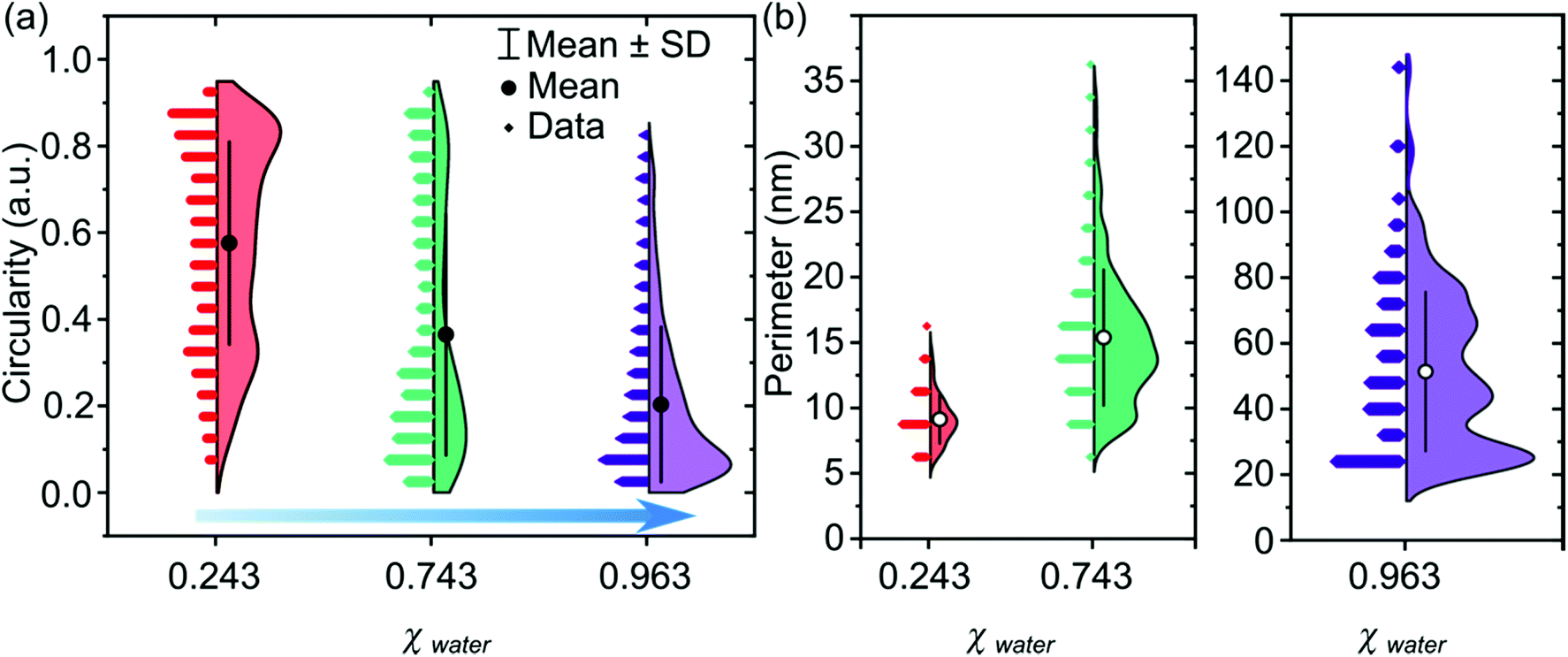
Controlling palladium morphology in electrodeposition from nanoparticles to dendrites via the use of mixed solvents - Nanoscale (RSC Publishing)
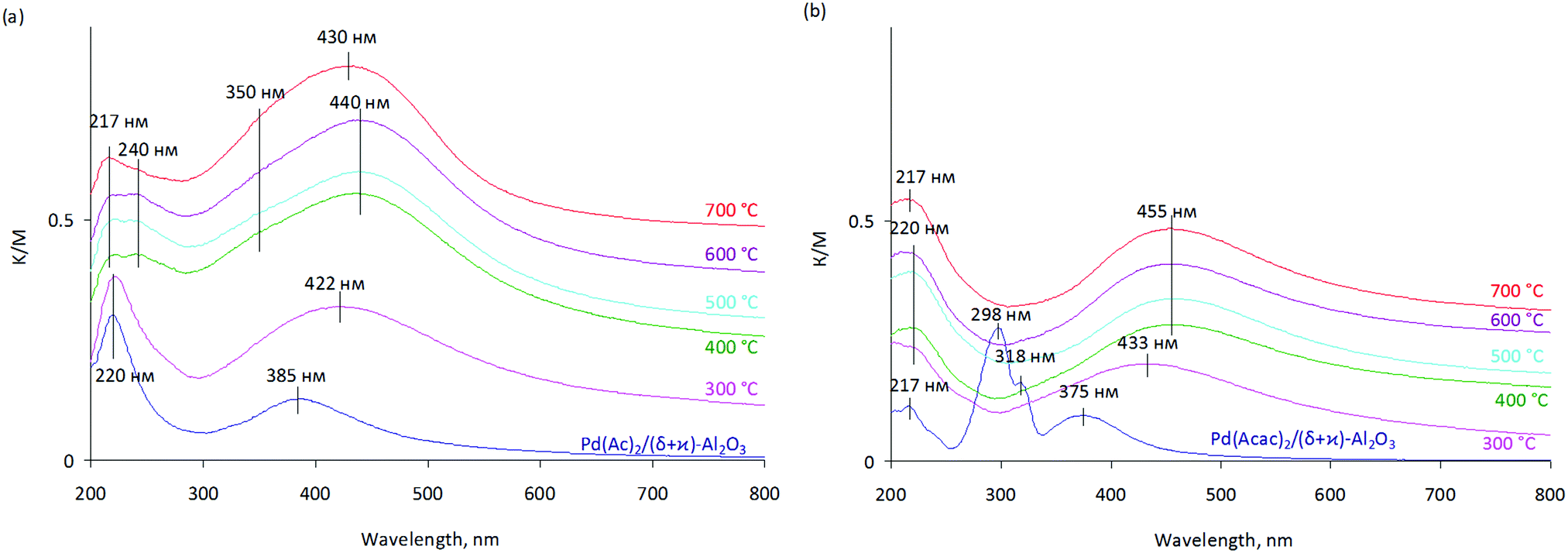
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)

![Effect of varying [KNO 3 ] and [KCl] on the rate of oxidation of D-fru... | Download Table Effect of varying [KNO 3 ] and [KCl] on the rate of oxidation of D-fru... | Download Table](https://www.researchgate.net/profile/Yokraj_Katre/publication/275552100/figure/tbl3/AS:669689210474507@1536677758457/Effect-of-varying-KNO-3-and-KCl-on-the-rate-of-oxidation-of-D-fru-at-303-K.png)

